Paschen Curve
Contents
Description of the Experiment
Being a different state of matter, there must be conditions at which a gaseous material progresses into plasma state. This experiment explores the conditions at which this transition occurs by studying the breakdown voltage, that is the voltage at which the plasma is formed, between parallel plates as a function of pressure and the distance between plates. Since the plasma is a good conductor, unlike the gas, it is fairly easy to verify this transition by watching the current drawn from power supply.
Links
- Video: rtsp://elabmc.ist.utl.pt/paschen.sdp
- Laboratory: Advanced in e-lab.ist.utl.pt[1]
- Control room: Paschen Curve
- Level: ****
Experimental Apparatus
The state of matter transition from gas into plasma is investigated in this experiment, allowing the study of Paschen's law. This is accomplished through an apparatus similar to the one used by Paschen, in which a voltage is applied to two parallel electrodes surrounded by low pressure gas. The breakdown voltage is determined as a function of pressure and distance separating the two plates.
Protocol
The breakdown phenomenon can be interpreted as a chain reaction, where one charged particle collides with a neutral one and generates an extra charged particle. If this collision process has a net gain, then there will be a discharge across the electrodes, otherwise the process will decay and the ionization will stop. It is therefore required that energy of the impacting particle exceeds the ionization energy of the neutral particle, so that there is no recombination and this is a function of both the gas pressure and the plates distance.
Using the model for a chain reaction, it can be shown that the breakdown voltage under certain conditions is described by the equation:
[math] V = \dfrac{a.pd}{\ln(pd) + b} [/math]
Where (i) \(V\) is the breakdown voltage, (ii) \(p\) is the pressure, and (iii) \(d\) is the gap distance. The constants (iv) \(a\) and (v) \(b\) depend upon the composition of the gas.
Taking into account the fact that there is no remote control over the distance between electrodes, the first step for the user should be to devise the pressure range through which the experiment will be performed. This will determine the number of times you will have to do the experiment. To configure this parameter, you must use the "Gas Pressure" slider. While the experiment is not in use, the vacuum pump will be connected to the main reaction chamber since the last time the experiment was performed until the moment it was activated. Considering that by the end of the experiment the pump down is monitored as it goes down until it reaches a limit of \(0.05 mBar\), the pressure inside the vessel before the experiment starts will always be at least that one. After the experiment begins, the chamber will be filled with Argon gas until the "Gas Pressure" is reached.
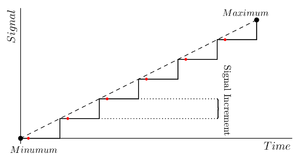
After that, the user can choose the voltage sweeping characteristics by selecting its "Maximum", "Minimum" and "Increase Step", which affect the signal as shown in figure 2. Choosing a bigger step might hide the details but will allow a faster determination of the region at which the breakdown occurs, while a smaller step will provide a higher detail on the determination of the data. It is recommended that the step value remains constant, since the quickly varying signal of the steps makes the breakdown easier and if it changes throughout the experiment the data will not be usable.
Finally the data acquisition is also configured by setting the "Maximum", "Minimum" and "Increase Step", since the ADCs performs a data acquisition every time the voltage is increased. Therefore the number of data points is equal to the number of times the voltage has to be updated.
As mentioned in the beginning, this setup performs the experiment under constant pressure, so this will only result in one data point. To get the Paschen Curve, the user should go through a large range of pressures.
Data Analysis
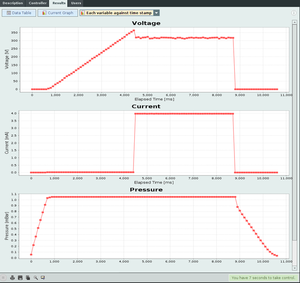
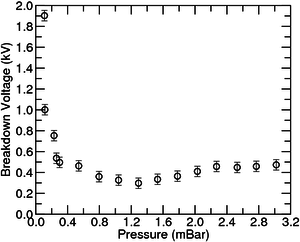
The data from a single experiment gives only one point, so the experiment should be repeated several times for different pressures. As with the other e-lab experiment, when the user activates the experiment and the pressure is set, a voltage ramp starts sweeping the electrodes, and the ADC is used to get both current and voltage applied to the electrodes. The software client prints the values in the interface of the voltage, current and pressure during the whole experiment. After that, the user will be able to see a clear transition from 0 to saturation in the current graph. The corresponding point in the voltage is that pressure breakdown similar to the contents of figure 3. After gathering the points of many different pressures, the user will have a data set similar to the one displayed in figure 4. Although there is a very high precision in the determination of the voltage values in the chamber, multiple runs of the experiment under the same conditions will show that there is often a window of about 50V under which the disruption can occur. Therefore this value was used for errors in the fit instead of the error with the ADC.
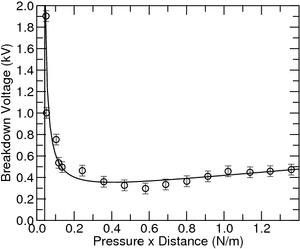
As it is, the data is still in raw form. The values in pressure must be multiplied by the gap distance and only after this the experimental data is ready to be adjusted to the equation 1, as seen in figure 5. In order to adjust to a systematic error in the measurements, you should add an extra fitting parameter:
[math] V = \dfrac{a.(pd + c)}{\ln(pd + c) + b} [/math]
From the fit \(a\) and \(b\) can be extracted, which can allow the determination of the gas inside the chamber. On the contrary, taking the information regarding the gas inside the chamber, in this case Argon, the data points can be used to determine the distance between the plates by fitting the data to the equation 2.
Links
- Principles of Plasma Diagnostics, Hutchinson, Cambridge
- Portuguese version (Versão em Português)