Determination of Planck's Constant
Description of the Experiment
In this experiment is possible study the photoelectric effect and calculate Planck's constant. Using 5 colored leds and a photoelectric cell.
Experimental Apparatus
The photoelectric cell is from the apparatus PASCO AP-9368. It works like a capacitor where an electric conductor emits photoelectrons. The potential of the photocell will increase with the emission of photoelectrons. And the stop potential will depend on the wavelength of the incident light, photoelectric effect.
The photocell is connected to ground to discharge, after each use.
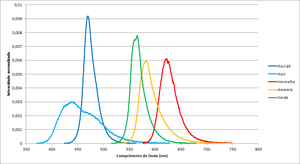
The leds have different spectrum and intensities so the time to reach the stop potencial may vary.
| Color | Frequency (THz) | Wavelegth (nm) | Espectros dos leds |
|---|---|---|---|
| Blue.ab | 638.7 | 469.70 | File:Espectro Azul.ab.txt |
| Blue | 684.6 | 438.20 | File:Espectro Azul.txt |
| Red | 482.2 | 622.21 | File:Espectro Vermelho.txt |
| Yellow | 514.4 | 583.16 | File:Example.txt |
| Green | 530.8 | 565.22 | File:Espectro Verde.txt |
Protocol
The number of photoelectrons emited will increase with the intensity of light. (corpuscular behavior of light)
- Choose a led to light up on the photocell
- Measure the stopping potential. Take note the time necessary to reach the maximum potential.
- Repit step 2 for diferent intensities.
| Color #1 __________(name) | Intensity (%) | Stop Potential (V) | Time (s) |
|---|---|---|---|
| 100 | |||
| 80 | |||
| 60 | |||
| 40 | |||
| 20 |
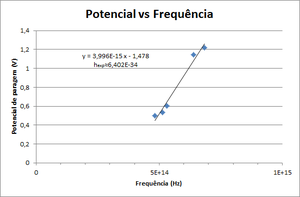
The photoelectron's kinetic energy depend only on the frequency of light. If the frequency of light increase the energy will increase.
- Obtain the stop potentials for different color leds.
- Draw a graphic of Stop Potential vs Frequency. Fit it to \( V = \frac{h}{e} \times \nu - \frac{W_0}{e} \) and obtain Planck's constant.
| Color (name) | Frequency (Hz) | Stop Potencial (V) |
|---|---|---|